UNIST site map
- Admissions
-
Academics
- Colleges and Schools
-
Academic Affairs
- Academic Calendar
- Academic Curriculum
- Requirements for Graduation
- Browse Open Courses
- Undergraduate Administration
-
Graduate Academic Affairs
- Tuition Fee Payment
- Academic Leave of Absence/ Academic Return
- Voluntary Withdrawal/ Expulsion
- Change of Major
- Change of Degree Program/ Dropping of Degree Program
- Class Period/ Attendance Period/ Academic Year・Semester
- Course Registration
- Course Drop
- Attendance/ Grade/ Exam
- Credit Transfer/ Credit Carryover
- Academic Forms
- Education Support
-
Research/Industry
- Research Aims
- Research Findings
- Researcher Search
-
Research Organizations
- UNIST Multi-Interdisciplinary Institute
- IBS Research Groups
-
UNIST Labs
- Department of Mechanical Engineering
- School of Energy and Chemical Engineering
- Department of Civil
- Department of Materials Science and Engineering
- Department of Nuclear Engineering
- Department of Industrial Engineering
- Department of Design
- Department of Biomedical Engineering
- Department of Biological Sciences
- Department of Electrical Engineering
- Department of Computer Science and Engineering
- Department of Mathematical Sciences
- Department of Chemistry
- Department of Physics
- School of Business Administration
- Graduate School of Carbon Neutrality
- Graduate School of Artificial Intelligence
- Research Support
- University-Industry Relations
- Campus Life
- News Center
- About UNIST
-
etc
- UNIST Bulletin
- Work-Life Balance Support System
- UNIST Gender Equality Plan
- Faculty Invitation for Tenure Track
- Faculty Invitation for Non-Tenure Track
- Board Meeting Minutes
- University Council Meeting Minutes
- Administrative Service Charter
- Privacy Policy
- Copyright Policy
- Rejection of Unauthorized Email Collection
- Operation and Management Policy for Video Information Processing Devices
- Information Disclosure


Connection Points of Knowledge, Everything About UNIST
Try searching.
Recommended search terms


- portal
- U Academics Innovation Center
- Leadership Center
- Dormitory
- Academic Information Center
- International Students Support
- Browse Open Courses
- Course Registration
- Graduation Requirements for Graduation
- Academic Leave of Absence/ Academic Return
- Military Service
- Certificate Issuance
- Academic Calendar
- Scholarships
- Campus Map
- Campus Life Guidebook
- Health Care Center
- Human Rights Center
- portal
- Job Opening
- Announcement for Bid
- UNIST AI Services
- UNIST Daycare Center
- Sports Center
- UI Downloads
- Announcement
- Recruitment of Professors (Non-tenure)
- Faculty Invitation for Tenure Track
- UNIST Academic Information Center
- Office of Research Facilities and Training
- Office of Research Affairs
- Rule Management System
- Academic Calendar


NEWS CENTER
Discover not only Research Findings and event news, but also the diverse facets of UNIST presented by reporters and writers.
News Center
UNIST News
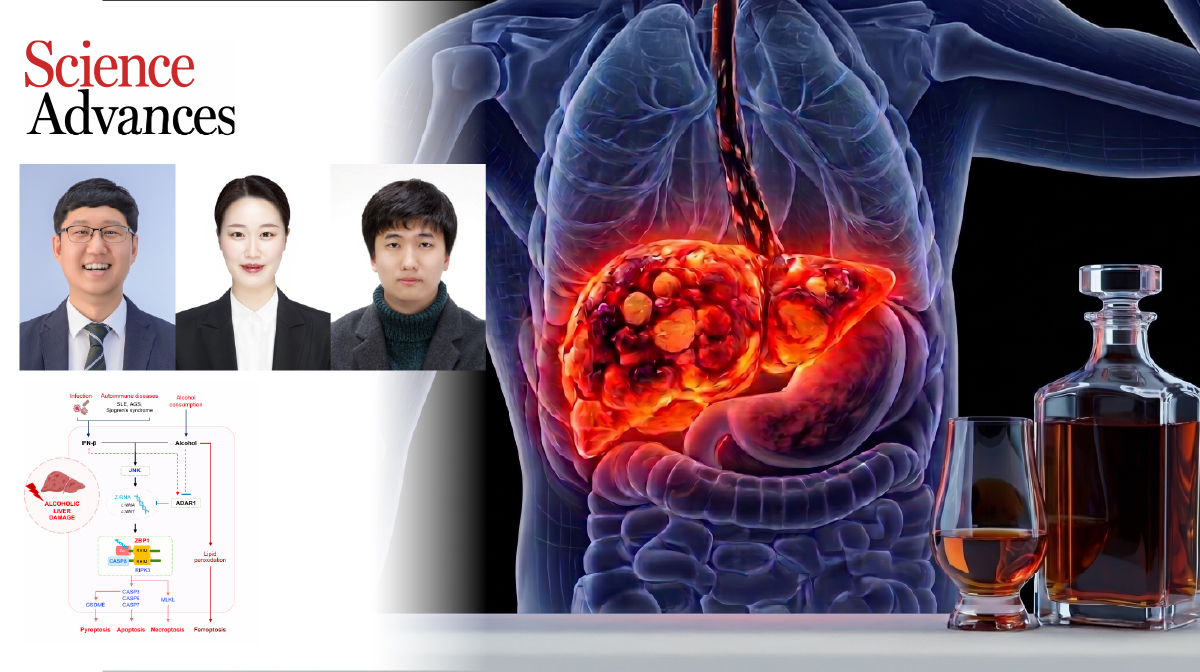
Breakthrough Unveils How Alcohol Amplifies Liver Damage During Illness
The findings of this research were published in Science Advances on April 10, 2026.
- Research
- JooHyeon Heo
- 2026.04.24
- 26
Abstract
A research team, affiliated with UNIST has uncovered a novel molecular pathway explaining why alcohol consumption can lead to severe liver damage, particularly when individuals are already battling infections, such as cold and influenza.
Led by Professor SangJoon Lee from the Department of Biological Sciences at UNIST, alongside Professors Rajendra Karki of Seoul National University and Si Ming Man of the Australian National University, the research sheds light on how alcohol interacts with the immune system to accelerate liver cell death and exacerbate liver disease.
The team discovered that, in the presence of interferon—a key immune signaling molecule produced during infections—alcohol triggers an inflammatory cascade within liver cells. Specifically, alcohol increases the level of Z-RAN, an abnormal form of RNA, which is detected by the pattern recognition receptor Z-DNA binding protein 1 (ZBP1). This activation initiates inflammatory cell death, contributing to liver damage.
Under normal circumstances, the protein ADAR1 suppresses Z-RNA formation, preventing unwarranted immune activation. However, the study revealed that alcohol impairs ADAR1 production, allowing Z-RNA to accumulate and activate ZBP1. This process involves the JNK signaling pathway, which was validated through animal experiments showing that inhibiting ZBP1 or blocking JNK signaling significantly reduced liver injury—even in the presence of alcohol and interferon.
Professor Lee explained, "While the direct toxicity of alcohol on liver cells has long been recognized, our findings highlight an immune-mediated pathway that exacerbates liver damage during illness. This discovery opens new possibilities for targeted therapies, such as ZBP1 inhibitors, to treat alcohol-related liver diseases."
The findings of this research were published in Science Advances on April 10, 2026. The study was supported by the National Research Foundation of Korea (NRF), the Korea Drug Development Find (KDDF), the Institute for Basic Science (IBS), the Circle Foundation, and Yuhan Corporation. It has also been supported through the Global Physician-Scientist Training Program and the ARPHA-H Project from the Korea Health Industry Development Institute (KHIDI) under the Ministry of Health and Welfare (MOHW).
Journal Reference
Yeonseo Jang, Hoeun Bae, SuHyeon Oh, et al ., “Innate immune sensing of dietary alcohol ignites inflammation to drive alcohol-related disease,” Adv. Sci ., (2026).
Related Links