UNIST site map
- Admissions
-
Academics
- Colleges and Schools
-
Academic Affairs
- Academic Calendar
- Academic Curriculum
- Requirements for Graduation
- Browse Open Courses
- Undergraduate Administration
-
Graduate Academic Affairs
- Tuition Fee Payment
- Academic Leave of Absence/ Academic Return
- Voluntary Withdrawal/ Expulsion
- Change of Major
- Change of Degree Program/ Dropping of Degree Program
- Class Period/ Attendance Period/ Academic Year・Semester
- Course Registration
- Course Drop
- Attendance/ Grade/ Exam
- Credit Transfer/ Credit Carryover
- Academic Forms
- Education Support
-
Research/Industry
- Research Aims
- Research Findings
- Researcher Search
-
Research Organizations
- UNIST Multi-Interdisciplinary Institute
- IBS Research Groups
-
UNIST Labs
- Department of Mechanical Engineering
- School of Energy and Chemical Engineering
- Department of Civil
- Department of Materials Science and Engineering
- Department of Nuclear Engineering
- Department of Industrial Engineering
- Department of Design
- Department of Biomedical Engineering
- Department of Biological Sciences
- Department of Electrical Engineering
- Department of Computer Science and Engineering
- Department of Mathematical Sciences
- Department of Chemistry
- Department of Physics
- School of Business Administration
- Graduate School of Carbon Neutrality
- Graduate School of Artificial Intelligence
- Research Support
- University-Industry Relations
- Campus Life
- News Center
- About UNIST
-
etc
- UNIST Bulletin
- Work-Life Balance Support System
- UNIST Gender Equality Plan
- Faculty Invitation for Tenure Track
- Faculty Invitation for Non-Tenure Track
- Board Meeting Minutes
- University Council Meeting Minutes
- Administrative Service Charter
- Privacy Policy
- Copyright Policy
- Rejection of Unauthorized Email Collection
- Operation and Management Policy for Video Information Processing Devices
- Information Disclosure


Connection Points of Knowledge, Everything About UNIST
Try searching.
Recommended search terms


- portal
- U Academics Innovation Center
- Leadership Center
- Dormitory
- Academic Information Center
- International Students Support
- Browse Open Courses
- Course Registration
- Graduation Requirements for Graduation
- Academic Leave of Absence/ Academic Return
- Military Service
- Certificate Issuance
- Academic Calendar
- Scholarships
- Campus Map
- Campus Life Guidebook
- Health Care Center
- Human Rights Center
- portal
- Job Opening
- Announcement for Bid
- UNIST AI Services
- UNIST Daycare Center
- Sports Center
- UI Downloads
- Announcement
- Recruitment of Professors (Non-tenure)
- Faculty Invitation for Tenure Track
- UNIST Academic Information Center
- Office of Research Facilities and Training
- Office of Research Affairs
- Rule Management System
- Academic Calendar


NEWS CENTER
Discover not only Research Findings and event news, but also the diverse facets of UNIST presented by reporters and writers.
News Center
UNIST News
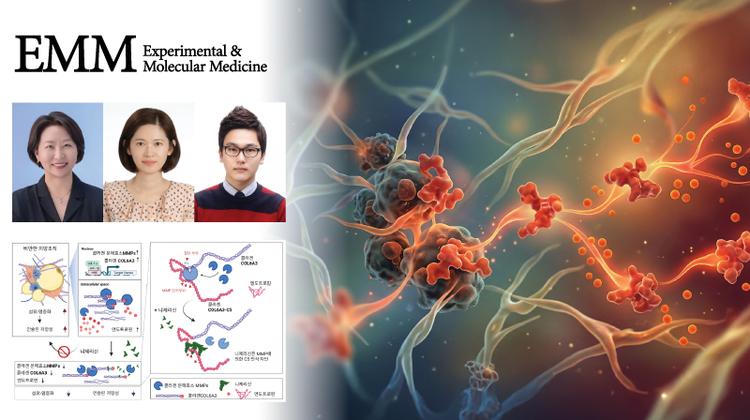
New Study Reveals Key Protein's Role in Regulating Dopamine Secretion!
Published in the November 2023 issue of Experimental & Molecular Medicine (EMM), their findings shed light on dopamine-related brain disease.
- Research
- JooHyeon Heo
- 2023.11.20
- 4019
Abstract Dopamine neurons are essential for voluntary movement, reward learning, and motivation, and their dysfunction is closely linked to various psychological and neurodegenerative diseases. Hence, understanding the detailed signaling mechanisms that functionally modulate dopamine neurons is crucial for the development of better therapeutic strategies against dopamine-related disorders. Phospholipase Cγ1 (PLCγ1) is a key enzyme in intracellular signaling that regulates diverse neuronal functions in the brain. It was proposed that PLCγ1 is implicated in the development of dopaminergic neurons, while the physiological function of PLCγ1 remains to be determined. In this study, we investigated the physiological role of PLCγ1, one of the key effector enzymes in intracellular signaling, in regulating dopaminergic function in vivo. We found that cell type-specific deletion of PLCγ1 does not adversely affect the development and cellular morphology of midbrain dopamine neurons but does facilitate dopamine release from dopaminergic axon terminals in the striatum. The enhancement of dopamine release was accompanied by increased colocalization of vesicular monoamine transporter 2 (VMAT2) at dopaminergic axon terminals. Notably, dopamine neuron-specific knockout of PLCγ1 also led to heightened expression and colocalization of synapsin III, which controls the trafficking of synaptic vesicles. Furthermore, the knockdown of VMAT2 and synapsin III in dopamine neurons resulted in a significant attenuation of dopamine release, while this attenuation was less severe in PLCγ1 cKO mice. Our findings suggest that PLCγ1 in dopamine neurons could critically modulate dopamine release at axon terminals by directly or indirectly interacting with synaptic machinery, including VMAT2 and synapsin III.
A groundbreaking study led by Professor Jae-Ick Kim from the Department of Biological Sciences at UNIST has uncovered a novel mechanism for controlling dopamine secretion. The research, published in the prestigious journal Experimental & Molecular Medicine, highlights the role of phospholipase Cγ1 (PLCγ1), a crucial protein involved in intracellular signaling, in regulating dopamine release.
Dopamine, a neurotransmitter essential for voluntary movement, reward learning, and motivation, is closely tied to various psychological and neurodegenerative disorders. Understanding the intricate signaling mechanisms that modulate dopamine neurons is vital for the development of effective therapeutic strategies against dopamine-related diseases.
 Figure 1. Genetic deletion of PLCγ1 enhances the release of dopamine at dopaminergic terminals.
Figure 1. Genetic deletion of PLCγ1 enhances the release of dopamine at dopaminergic terminals.
Previous studies had suggested that PLCγ1 may be implicated in the development of dopaminergic neurons. However, the precise physiological function of PLCγ1 remained elusive. To address this knowledge gap, Professor Kim's team conducted an in-depth investigation into the role of PLCγ1 in regulating dopaminergic function in vivo.
The study utilized cell type-specific deletion of PLCγ1 in dopamine neurons to evaluate its impact on dopamine release. Surprisingly, the deletion of PLCγ1 did not adversely affect the development and cellular morphology of midbrain dopamine neurons. Instead, it led to a notable increase in dopamine release from dopaminergic axon terminals in the striatum.
The enhanced dopamine release observed in the study was accompanied by an upregulation of vesicular monoamine transporter 2 (VMAT2) at dopaminergic axon terminals. VMAT2 is responsible for transporting dopamine into synaptic vesicles. Additionally, the knockout of PLCγ1 in dopamine neurons resulted in heightened expression and colocalization of synapsin III, a protein that controls the trafficking of synaptic vesicles.
Furthermore, the study demonstrated that the knockdown of both VMAT2 and synapsin III in dopamine neurons significantly attenuated dopamine release. However, this attenuation was less severe in PLCγ1 knockout mice, suggesting that PLCγ1 critically modulates dopamine release at axon terminals by directly or indirectly interacting with synaptic machinery.
 Figure 2. Altered synapsin III expression might underlie the increased localization of VMAT2 in the dopaminergic axons of PLCγ1 cKO mice. (A) Representative confocal images of VMAT2 expression in striatal dopaminergic axons (white arrowhead: colocalized area). (B) VMAT2 and TH colocalized area and the percentage of VMAT-positive area on TH-positive axons. (C) Representative confocal images showing the expression of synapsin III in striatal dopaminergic axons (white arrowhead: colocalized area). (D) Synapsin III and TH colocalized area in the DStr and the percentage of synapsin III-positive area on TH-positive axons in the DStr.
Figure 2. Altered synapsin III expression might underlie the increased localization of VMAT2 in the dopaminergic axons of PLCγ1 cKO mice. (A) Representative confocal images of VMAT2 expression in striatal dopaminergic axons (white arrowhead: colocalized area). (B) VMAT2 and TH colocalized area and the percentage of VMAT-positive area on TH-positive axons. (C) Representative confocal images showing the expression of synapsin III in striatal dopaminergic axons (white arrowhead: colocalized area). (D) Synapsin III and TH colocalized area in the DStr and the percentage of synapsin III-positive area on TH-positive axons in the DStr.
Professor Kim explained, "Our study provides novel insights into the signaling mechanisms within dopamine neurons, which were previously challenging to decipher. By identifying the role of dopamine neuron-specific PLCγ1, we have paved the way for potential advancements in the treatment of dopamine-related brain diseases."
Hye Yun Kim, the first author of the study, expressed optimism regarding the future impact of their findings. "By elucidating the PLCγ1-mediated signaling pathways involved in the regulation of dopamine secretion, we hope to contribute to the development of innovative treatments for dopamine-related brain diseases."
The research was conducted in collaboration with the Korea Brain Research Institute (KBRI) and received support from the Mid-Career Researcher Program and the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Science and ICT (MSIT).
Journal Reference
Hye Yun Kim, Jieun Lee, Hyun-Jin Kim, et al., "PLCγ1 in dopamine neurons critically regulates striatal dopamine release via VMAT2 and synapsin III," EMM, (2023).
Related Links